|
As a result, a number of methods have been developed to expand the utility of STED and to allow more information to be provided.įrom the beginning of the process, STED has allowed fluorescence microscopy to perform tasks that had been only possible using electron microscopy. Over the last several years, STED has developed from a complex and highly specific technique to a general fluorescence method. In addition, Atto 647N was first used with this method to produce two-color STED. 
Since that first application, this technique has been applied to a much wider range of dyes including green emitting, Atto 532, and yellow emitting, Atto 590, as well as additional red emitting dyes. The first use of this technique coupled MR-121SE, a red dye, with a secondary anti-mouse antibody. These cells are STED fluorescent dyes bound to antibodies through amide bonds. One such advancement was the development of immunolabeled cells. This desire for better analysis of these systems has led to living cell STED and multicolor STED, but it has also demanded more and more advanced dyes and excitation systems to accommodate the increased functionality. To allow for STED analysis of biological systems, the dyes and laser sources must be tailored to the system. As a result, the first dyes used were laser emitting in the red spectrum. Rhodamine B was named in the first theoretical description of STED. That imposes certain constraints on the optics that can be used.Įarly on in the development of STED, the number of dyes that could be used in the process was very limited. To optimize the effectiveness of STED, the destructive interference in the center of the focal spot needs to be as close to perfect as possible. The equation is:ĭ = λ 2 n sin α = λ 2 N A. Ernst Abbe developed an equation to describe this limit. In traditional microscopy, the resolution that can be obtained is limited by the diffraction of light. The double laser design allows for excitation and stimulated emission to be used together for STED. This redshift allows the stimulated photon to be ignored. Jablonski diagram showing the redshift of the stimulated photon. This differs from the stochastic functional techniques such as photoactivated localization microscopy (PALM) and stochastic optical reconstruction microscopy (STORM) as these methods use mathematical models to reconstruct a sub diffraction limit from many sets of diffraction limited images.īackground Ernst Abbe's formula for the diffraction limit, set in stone at a monument in Jena. STED is a deterministic functional technique that exploits the non-linear response of fluorophores commonly used to label biological samples in order to achieve an improvement in resolution, that is to say STED allows for images to be taken at resolutions below the diffraction limit. STED microscopy is one of several types of super resolution microscopy techniques that have recently been developed to bypass the diffraction limit of light microscopy to increase resolution. This patent was unknown to Hell and Wichmann in 1994. 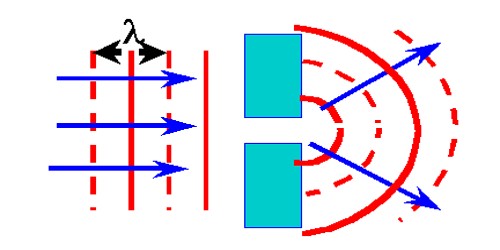
Okhonin (Institute of Biophysics, USSR Academy of Sciences, Siberian Branch, Krasnoyarsk) had patented the STED idea. Hell was awarded the Nobel Prize in Chemistry in 2014 for its development. Hell and Jan Wichmann in 1994, and was first experimentally demonstrated by Hell and Thomas Klar in 1999. It creates super-resolution images by the selective deactivation of fluorophores, minimizing the area of illumination at the focal point, and thus enhancing the achievable resolution for a given system. Stimulated emission depletion ( STED) microscopy is one of the techniques that make up super-resolution microscopy. Technique in fluorescence microscopy Stimulated emission depletion (STED) microscopy provides significant resolution improvements over those possible with Confocal microscopy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
